Run. Analyse. Improve.
TEMPLO® Running Analysis
Professional solution for markerless Running Analysis
Capture the running movement with high-speed cameras to visualise movement patterns frame by frame.
Body points are automatically captured via AI to objectify the analysis and report data. Use predefined analysis templates and an intuitive user interface for practical and time-efficient use in everyday life.

By loading the video, you agree to YouTube's privacy policy.
Learn more
Reference Projects



Discover the features
evidence-based
utmost flexibility
streamlined workflow
meaningful reporting

By loading the video, you agree to YouTube's privacy policy.
Learn more
2D Analysis
Record the running style of your athlete or customer without markers and benefit from intelligent and fast data acquisition and evaluation.
Evaluate the automatically generated joint angles and assess the movement of the anatomical reference points which are overlayd on the video image.
3D Running analysis at the highest level
By integrating additional cameras, you can analyse the running movement in 3D. The fully automatic evaluation of the analysis provides comprehensive results on all relevant running parameters.

By loading the video, you agree to YouTube's privacy policy.
Learn more

By loading the video, you agree to YouTube's privacy policy.
Learn more
Save analysis time
The fully automated motion capture gives you the possibility to record all relevant motion data within a few minutes without markers. Save valuable analysis time and gain measurement accuracy. With a single click, the processed tracking data from your recorded running analysis is displayed.
Make results visible
Use our TEMPLO® software to visualise the analysis results for your athletes and patients. Present your results immediately after recording with the help of our analysis tools. Export videos and create predesigned or customised reports to share with athletes, clients, coaches or therapists.
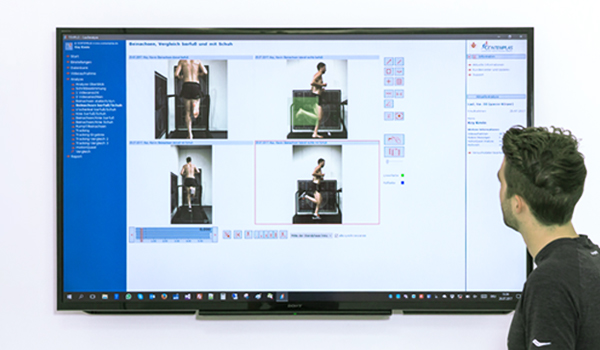

Analyse comprehensively
Expand your analysis with additional measurement data from force plates, EMG and complementary timing methods and obtain a comprehensive biomechanical view of your athletes and clients individual running style.
Running Analysis in use

By loading the video, you agree to Vimeo's privacy policy.
Learn more
CHOOSE YOUR INDIVIDUAL
Running Analysis System
TEMPLO® RUNNING ANALYSIS offers you all possibilities. From simple recording to markerless tracking.
Do you need help with the selection? Contact us.

SET UP
The TEMPLO® RUNNING ANALYSIS consists of the following components:
BASIC
2 Highspeed Cameras [1]
Notebook or PC with software [2]
ADD ONS
Treadmill with integrated pressure measurement [3+4]
OptoGait [5]
EMG [6]
Lighting system [7]
Calibration board [8]
* No CONTEMPLAS GmbH product is classified as a medical device under MDR (EU) 2017/745 within the European Union, United Kingdom (excluding Northern Ireland) under Part II, UK MDR 2002 (SI 2002 no. 618) or within the requirements of any other national medical regulatory body. CONTEMPLAS GmbH supplied products must not be used where the intention is to provide information which will be used to take decisions with diagnosis or therapeutic purposes.
